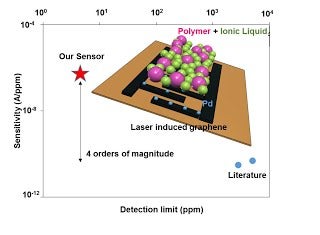
Methane is a potent greenhouse gas with large emissions occurring across gas distribution networks and mining/extraction infrastructure. The development of inexpensive, low-power electrochemical sensors could provide a cost-effective means to carry out distributed sensing to identify leaks for rapid mitigation. In this work, we demonstrate a simple and cost-effective strategy to rapidly prototype ultra-sensitive electrochemical gas sensors. A room temperature methane sensor is evaluated which demonstrates the highest reported sensitivity (0.55 mA/ppm/cm2) with a rapid response time (40 s) enabling sub-ppm detection. Porous, laser-induced graphene (LIG) electrodes are patterned directly into commercial polymer films and imbibed with a palladium nanoparticle dispersion to distribute the electrocatalyst within the high surface area support. A pseudo-solid state ionic liquid/polyvinylidene fluoride electrolyte was painted onto the flexible cell yielding a porous electrolyte, within the porous LIG electrode, simultaneously facilitating rapid gas transport and enabling the room temperature electro-oxidation pathway for methane. The performance of the amperometric sensor is evaluated as a function of methane concentration, relative humidity and tested against interfering gases.
2018
25. L. Xu, A.R. Tetreault, H.H. Khaligh, I.A. Goldthorpe, S.D. Wettig, M. A. Pope, "Continuous Langmuir-Blodgett deposition and transfer by controlled edge-to-edge assembly of floating 2D materials" Langmuir (2018 - In Press)
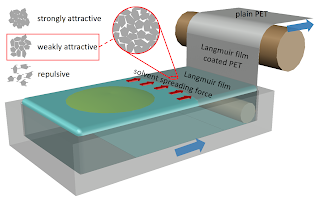
The Langmuir-Blodgett technique is one of the most controlled methods to deposit monomolecular layers of floating or surface active materials but has lacked the ability to coat truly large area substrates. In this work, by manipulating single layer dispersions of graphene oxide (GO) and thermally exfoliated graphite oxide (TEGO) into water-immiscible spreading solvents, unlike traditional Langmuir-Blodgett deposition which requires densification achieved by compressing barriers, we demonstrate the ability to control the 2D aggregation and densification behavior of these floating materials using a barrier-free method. This is done by controlling the edge-to-edge interactions through modified sub-phase conditions and by utilizing the distance-dependent spreading pressure of the deposition solvent. These phenomena allow substrates to be coated by continuous deposition and substrate withdrawal – enabling roll-to-roll deposition and patterning of large area substrates such as flexible polyethylene terephthalate. The aggregation and solvent-driven densification phenomena are examined by in situ Brewster angle video microscopy and by measuring the local spreading pressure induced by the spreading solvent acting on the floating materials using a Langmuir-Adam balance. As an example, the performance of films deposited in this way are assessed as passivation layers for Ag nanowire-based transparent conductors.
24. D. Ghosh, M. Gad, I. Lau, M. A. Pope, "Trapping and Redistribution of Hydrophobic Sulfur Sols in Graphene-Polyethyleneimine Networks for Stable Li-S Cathodes" Advanced Energy Materials 8 (2018), 1801979
The lithium–sulfur battery is a promising next-generation system which promises to be lower cost and more energy dense than current Li-ion technologies but only if cycle stability and sulfur utilization in thick, high areal loading cathodes could be realized. Herein we report a simple method to fabricate high capacity, high loading cathodes with one of the highest cycle-stabilities reported. We demonstrate that sulfur sols formed by crashing dissolved elemental sulfur into water are trapped between graphene oxide sheets when flocculated with polyethyleneimine. Low temperature, hydrothermal treatment produces a conductive, partially covalent composite exhibiting outstanding cycle-stability. Electrodes with high areal sulfur loadings (up to 5.4 mg/cm2) and fractions as high as 75.7 wt percent, achieved high capacity such that practical energy densities of > 400 Wh/kg could be realized. The electrodes demonstrate negligible capacity loss over 250 cycles at 0.15C and only 0.028 percent capacity loss per cycle over 810 cycles at 0.75C. Eventual capacity fading was found to be linked to degradation of the lithium-metal anode suggesting that the cathode material remained stable over even more extended cycling.
23. L. Dan, M. A. Pope, A. L. Elias, "Solution-Processed Conductive Biocomposites Based on Polyhydroxybutyrate and Reduced Graphene Oxide" Journal of Physical Chemistry C 122 (2018) 17490-17500
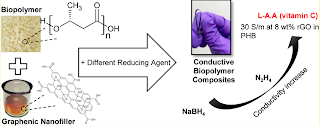
Graphenic material/biopolymer nanocomposites have attracted attention for use in next generation flexible and degradable electronics. However, achieving high electrical conductivity (above 10 S/m) and favorable mechanical properties in such materials remains a challenge. In this work, reduced graphene oxide (rGO)/polyhydroxybutyrate (PHB) films were both prepared from solution. Three reducing agents were investigated: sodium borohydride, hydrazine, and L-ascorbic acid. While for the first two reducing agents (sodium borohydride and hydrazine), GO was first reduced and then added to dissolved PHB, for the third reducing agent (L-ascorbic acid) an in-situ reduction was performed. We systematically investigated the impacts of the three reducing agents, by comparing their reduction efficiency, the residual functional groups presented on the GO, and the properties of the resulting composites of all the composites, reduction by L-ascorbic acid gives the lowest electrical percolation threshold (~1 wt%) and the highest electrical conductivity (30 S/m on 8 wt% loading). This conductivity value is on par with the highest known values for rGO/biopolymer composites. The mechanical properties of the composites were characterized; the ultimate tensile stress and Young's modulus of 4wt% composite formed with rGO reduced using either hydrazine or L-ascorbic acid were higher than for pure PHB. A strain sensor was demonstrated using these composites.
22. C.M. Tittle, D. Yilman, M.A. Pope, C.J. Backhouse, "Robust Superhydrophobic Laser-Induced Graphene for Desalination Applications" Advanced Materials Technologies (2018)
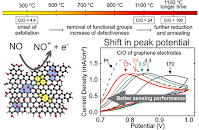
The fabrication of long-lived, superhydrophobic surfaces using a manufacturable process is an important challenge for material science. Significant advances have been reported; however, many surfaces suffer from fragility, non-manufacturable fabrication techniques, and temporal instability. Such challenges have limited commercial scale application of superhydrophobic films, including their application to water desalination where long lifetimes and durability are essential. We demonstrate the fabrication of controllably wettable surfaces formed from laser-induced graphene in atmospheric conditions with contact angle control from 59o to 176o; representing some of the most superhydrophobic carbon surfaces ever reported. This superhydrophobicity was used to engineer a membrane with the largest pores ever reported for the energy efficient water desalination technique of air-gapped membrane distillation. State-of-the-art production of distilled water was observed, and no membrane failure or loss of superhydrophobicity was observed on a time-scale of months - suggesting total water production capabilities well beyond anything yet demonstrated.
2017
21. Z. She, D. Ghosh, M.A. Pope, "Decorating Graphene Oxide with Ionic Liquid Nanodroplets: An Approach Leading to Energy Dense, High Voltage Supercapacitors" ACS Nano (2017 - Just Accepted)
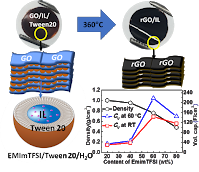
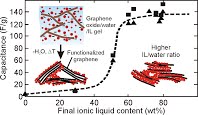
A major stumbling block in the development of high energy density graphene-based supercapacitors has been maintaining high ion-accessible surface area combined with high electrode density. Herein we develop an ionic liquid (IL)-surfactant microemulsion system which is found to facilitate the spontaneous adsorption of IL-filled micelles onto graphene oxide (GO). This adsorption distributes the IL over all available surface area and provides an aqueous formulation that can be slurry cast onto current collectors, leaving behind a dense nanocomposite film of GO/IL/surfactant. By removing the surfactant and reducing the GO through a low temperature (360 °C) heat treatment, the IL plays a dual role of spacer and electrolyte. We study the effect of IL content and operating temperature on the performance, demonstrating a record high gravimetric capacitance (302 F/g at 1 A/g) for 80 wt% IL composites. At 60 wt% IL, combined high capacitance and bulk density (0.76 g/cm3) yields one of the highest volumetric capacitances (218 F/cm3, at 1 A/g) ever reported for a high voltage IL-based supercapacitor While achieving promising rate performance and cycle-life, the approach also eliminates the long and costly electrolyte imbibition step of cell assembly since the electrolyte is cast directly with the electrode material.
20. A. Nasr Esfahani, A. Katbab, A. Taeb, L. Simon, M. A. Pope, "Correlation between mechanical dissipation and improved X-band electromagnetic shielding capabilities of amine functionalized graphene/thermoplastic polyurethane composites" European Polymer Journal 95 (2017) 520-538
Graphene-based polymer nanocomposites have demonstrated significant promise to create commercially viable electromagnetic interference (EMI) shielding to protect the next-generation of electronic materials from radiative pollution. In the present study, we carry out a systematic analysis of the dynamic mechanical, dielectric, electrical and X-band shielding properties of thermoplastic polyurethane (TPU) elastomer filled with amine functionalized graphene obtained by the rapid thermal expansion of graphite oxide. By preparation of nanocomposites based on modified and unmodified graphene using solution mixing and hot compression moulding, we demonstrate that the modification with 2-aminoethyl methacrylate enhances the EMI shielding from 14 to 25 dB. We also show by fracture analysis, cross-sectional transmission electron microscopy and dynamic mechanical analysis that the modification significantly strengthens the interfacial interactions between TPU and the functionalized graphene at the same filler loading. We find that the dominant shielding mechanism is through absorption and discuss the correlation between the viscoelastic mechanical loss tangent and the more effective dissipation of absorbed EM radiation which might account for the discrepancy between the theoretically predicted and experimentally observed EMI SE.
19. Y. Zhang, L. Xu, W.R. Walker, C.M. Tittle, C.J. Backhouse, M. A. Pope, "Langmuir films and uniform, large area, transparent coatings of chemically exfoliated MoS2 single layers" Journal of Materials Chemistry C (2017)
By manipulating colloidal dispersions of chemically exfoliated molybdenum disulfide (MoS2) into an appropriate spreading solvent, we demonstrate, for the first time, the ability to form stable, floating MoS2 Langmuir films without the use of surfactants or significant material loss into the aqueous sub-phase. While the floating sheets can be compressed into a densely tiled film by the barriers of a traditional Langmuir-Blodgett trough, we also report an edge-to-edge aggregation and spreading driven densification phenomena that allows the film to be built up from the outside of the trough inwards during the deposition process. Continued deposition allows us to fill the entire trough with a dense (85-95%) film of discretely tiled 1T MoS2 and to coat substrates as large as 130 cm2. The transfer efficiency is found to be as high as 120 m2 of coated area per gram of deposited MoS2. Comparing the transfer efficiency to the theoretical specific surface area of MoS2 provides a method to estimate film thickness and exfoliation efficiency. Atomic force microscopy and optical absorption measurements are used to corroborate this estimate of 2.8 layers for the traditional n-butyllithium exfoliation method used. We demonstrate that the films can be built up layer-by-layer and investigate the optical and electrical properties of the films before and after conversion from the 1T to 2H polymorph.
18. A. Azhari, E. Marzbanrad, D. Yilman, E. Toyserkani, M.A.Pope, "Binder-jet powder-bed additive manufacturing (3D printing) of thick graphene-based electrodes" 119 (2017) 257-266
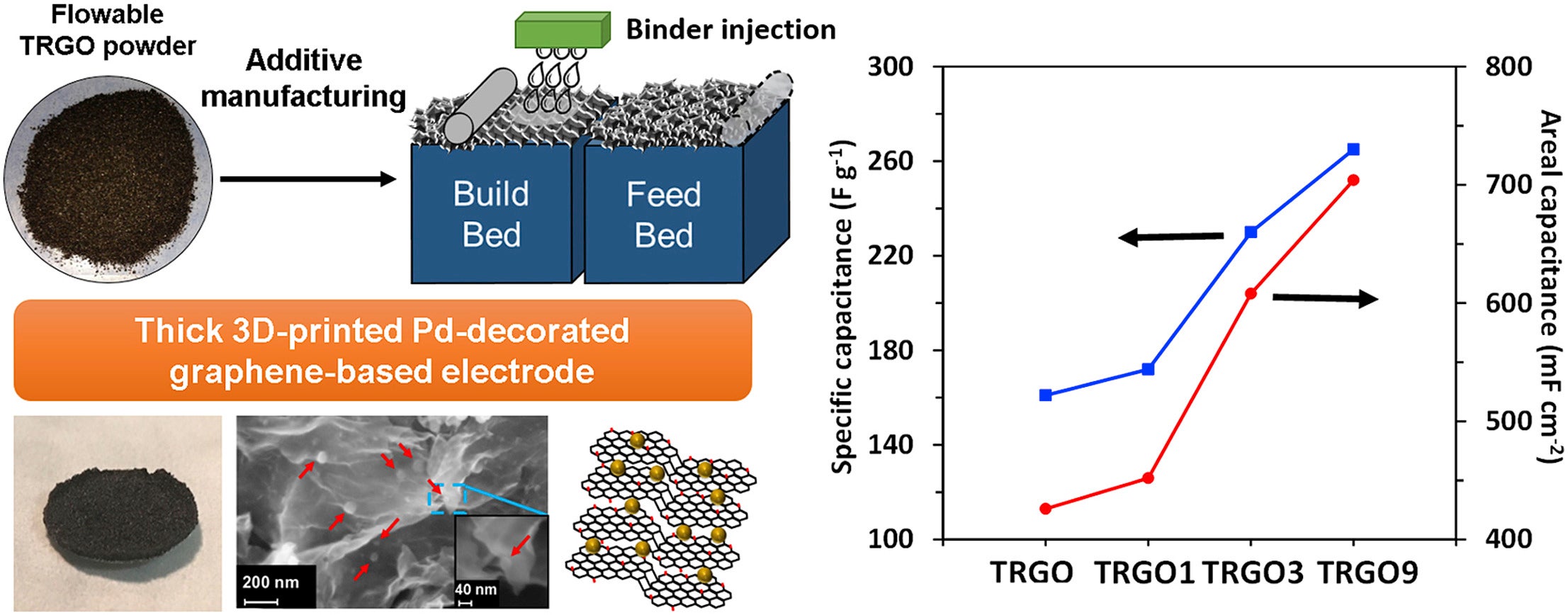
Additive manufacturing (AM), also known as 3D printing, is emerging as a promising method for the fabrication of complex 3D structures and has the potential to replace the conventional techniques used in the manufacture of commercial devices based on advanced materials. Graphene has shown superior performance in various electronic devices such as electrochemical supercapacitors. However, it remains challenging to produce the thick, high loading graphene-based electrodes required to achieve a high practical energy density in full devices. Herein, we introduce a powder-bed AM technique for the fabrication of crack-free, mm-thick graphene-based electrodes, with high surface area that can be printed in complex shapes. While this technology has the potential to be used in many application areas including energy storage, conversion, and sensing, in this work, we demonstrate their use as high performance supercapacitors. Devices fabricated using thermally exfoliated graphene oxide powder had gravimetric and areal capacitance of ∼260 F g−1 and ∼700 mF cm−2, respectively at five mV s−1 in 1 M H2SO4 electrolyte. The supercapacitors retained 80 percent of their capacity over 1000 cycles. This technique provides a promising route for the fabrication and commercialization of thick, porous graphene-based devices.
17. H. H. Khaligh, L Xu, A. Khosropour, A. Madeira, M. Romano,C. Pradére, M. Tréguer-Delapierre, L. Servant, M. A. Pope, I. A. Goldthorpe, "The Joule heating problem in silver nanowire transparent electrodes" Nanotechnology (2017)
Silver nanowire transparent electrodes have shown considerable potential to replace conventional transparent conductive materials. However, in this report we show that Joule heating is a unique and serious problem with these electrodes. When conducting current densities encountered in organic solar cells, the average surface temperature of indium tin oxide and silver nanowire electrodes, both with sheet resistances of 60 ohms/square, remains below 35 °C. However, in contrast to indium tin oxide, the temperature in the nanowire electrode is very non-uniform, with some localized points reaching temperatures above 250 °C. These hotspots accelerate nanowire degradation, leading to electrode failure after five days of continuous current flow. We show that graphene, a commonly used passivation layer for these electrodes, slows nanowire degradation and creates a more uniform surface temperature under current flow. However, the graphene does not prevent Joule heating in the nanowires and local points of high temperature ultimately shift the failure mechanism from nanowire degradation to melting of the underlying plastic substrate. In this paper, surface temperature mapping, lifetime testing under current flow, post-mortem analysis, and modelling illuminate the behavior and failure mechanisms of nanowires under extended current flow and provide guidelines for managing Joule heating.
2016
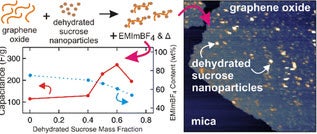
16. D. J. Bozym, S. Korkut, M.A. Pope, I.A. Aksay, "Dehydrated Sucrose Nanoparticles as Spacers for Graphene–Ionic Liquid Supercapacitor Electrodes" ACS Sustainable Chemistry & Engineering 4 (2016) 7167-7174
The addition of dehydrated sucrose nanoparticles increases the gravimetric capacitance of electrochemical double-layer capacitor electrodes produced via the evaporative consolidation of graphene oxide–water–ionic liquid gels by more than two-fold. Dehydrated sucrose adsorbs onto graphene oxide and serves as a spacer, preventing the graphene oxide from restacking during solvent evaporation. Despite 61 wt percent of the solids being electrochemically inactive dehydrated sucrose nanoparticles, the best electrodes achieved an energy density of ∼13.3 Wh/kg, accounting for the total mass of all electrode components.
15. M. Javidi, M.A. Pope, A.N. Hrymak, "Investigation on dip coating process by mathematical modeling of non-Newtonian fluid coating on cylindrical substrate" Physics of Fluids 28 (2016) 063105
A mathematical model for the dip coating process has been developed for cylindrical geometries with non-Newtonian fluids. This investigation explores the effects of the substrate radius and hydrodynamic behavior of the non-Newtonian viscous fluid on the resulting thin film on the substrate. The coatingfluid studied, Dymax 1186-MT, is a resin for fiber optics and used as a matrix to suspend 1 vol. percent titanium dioxide particles. The coating substrate is a 100 μm diameter fiber optic diffuser. Ellis viscosity model is applied as a non-Newtonian viscous model for coating thickness prediction, including the influence of viscosity in low shear rates that occurs near the surface of the withdrawal film.In addition, the results of the Newtonian and power law models are compared with the Ellis model outcomes. The rheological properties and surface tensionof fluids were analyzed and applied in the models and a good agreement between experimental and analytical solutions was obtained for Ellis model.
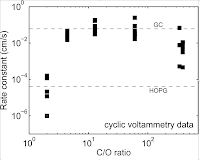
14. C. Punckt, M. Pope Y.M. Liu and I.A. Aksay, "Structure-Dependent Electrochemistry of Reduced Graphene Oxide Monolayers" Journal of the Electrochemical Society 163 (2016) H491-H498
While graphene and other carbonaceous nanomaterials have shown promise in a variety of electrochemical applications, measurement of their intrinsic performance is often confounded with effects related to the complexities due to diffusion in a porous medium. To by-pass this limitation, we use effectively non-porous tiled monolayers of reduced graphene oxide as a model platform to study how rates of heterogeneous electron transfer evolve as a function of graphene structure/chemistry. A variety of electrochemical systems are investigated including the standard ferri/ferrocyanide redox probe, several common biomolecular redox systems as well as copper electrodeposition. We show that the rates of heterogeneous electron transfer can vary by as much as three orders of magnitude depending on the reduction or annealing conditions used and the redox system investigated. Performance changes are linked to graphene chemistry, and we show that the graphene oxide reduction procedure must be chosen judiciously to maximize the electrochemical performance for particular applications.
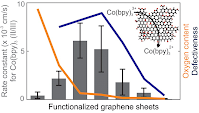
13. J.D. Roy-Mayhew, M.A. Pope, C. Punckt and I.A. Aksay, "Intrinsic Catalytic Activity of Graphene Defects for the CoII/III(bpy)3 Dye-Sensitized Solar Cell Redox Mediator" ACS Applied Materials & Interfaces 8 (2016) 9134–9141
We demonstrate that functionalized graphene, rich with lattice defects but lean with oxygen sites, catalyzes the reduction of CoIII(bpy)3 as well as platinum does, exhibiting a rate of heterogeneous electron transfer, k0, of ∼6 × 10–3 cm/s. We show this rate to be an order of magnitude higher than on oxygen-site-rich graphene oxide, and over 2 orders of magnitude higher than on the basal plane of graphite (as a surrogate for pristine graphene). Furthermore, dye-sensitized solar cells using defect-rich graphene monolayers perform similarly to those using platinum nanoparticles as the catalyst.
2015
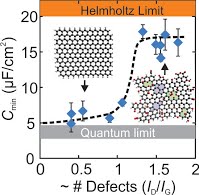
12. M.A. Pope and I.A. Aksay, "Four-fold Increase in the Intrinsic Capacitance of Graphene through Functionalization and Lattice Disorder." Journal of Physical Chemistry C 119 (2015) 20369–2037
Graphene has been heralded as a promising electrode material for high energy and power density electrochemical supercapacitors. This is in spite of recent work confirming the low intrinsic capacitance of the graphene/electrolyte interface limited by graphene’s low quantum capacitance, an effect known for the basal-plane of graphite for over four decades. Consistent with this limit, much of the supercapacitor research implies the use of pristine graphene but, in contrast, uses a functionalized and defective graphene formed through the reduction of graphene oxide, without clarifying why reduced graphene oxide is needed to achieve high capacitance. Herein, we show that an optimal level of functionalization and lattice disorder in reduced graphene oxide yields a four-fold increase in intrinsic capacitance over that of pristine graphene, suggesting graphene-based materials can indeed be tailored to engineer electrodes with significantly higher gravimetric capacitance limits exceeding 450 F/g than what has been achieved (~274 F/g) thus far, in non-aqueous electrolytes capable of high voltage operation.
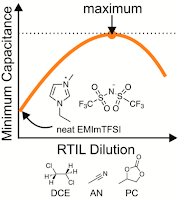
11. D.J. Bozym, B. Uralcan, D. Limmer, M.A. Pope, N.J. Szamreta, P.G. Debenedetti, I.A. Aksay, "Anomalous Capacitance Maximum of the Glassy Carbon-Ionic Liquid Interface through Dilution with Organic Solvents." Journal of Physical Chemistry Letters (2015 - Just Accepted)
We use electrochemical impedance spectroscopy to measure the effect of diluting a hydrophobic room temperature ionic liquid with miscible organic solvents on the differential capacitance of the glassy carbon–electrolyte interface. We show that the minimum differential capacitance increases with dilution and reaches a maximum value at ionic liquid contents near five to 10 mol% (i.e., ∼1 M). We provide evidence that mixtures with 1,2-dichloroethane, a low-dielectric constant solvent, yield the largest gains in capacitance near the open circuit potential when compared against two traditional solvents, acetonitrile and propylene carbonate. To provide a fundamental basis for these observations, we use a coarse-grained model to relate structural variations at the double layer to the occurrence of the maximum. Our results reveal the potential for the enhancement of double-layer capacitance through dilution.
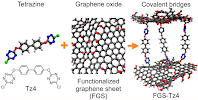
10. Y. Li , V. Alain-Rizzo , L. Galmiche , P. Audebert , F. Miomandre , G. Louarn , M. Bozlar , M. A. Pope , D. M. Dabbs, I. A. Aksay, "Functionalization of Graphene Oxide by Tetrazine Derivatives: A Versatile Approach toward Covalent Bridges between Graphene Sheets." Chemistry of Materials (2015 - Just Accepted)
We have covalently grafted tetrazine derivatives to graphene oxide through nucleophilic substitution. Since the tetrazine unit is electroactive and nitrogen-rich, with a reduction potential sensitive to the type of substituent and degree of substitution, we used electrochemistry and Xray photoelectron spectroscopy to demonstrate clear evidence for grafting through covalent bonding. Chemical modification was supported by Fourier transform infrared spectroscopy and thermal analysis. Tetrazines grafted onto graphene oxide displayed different mass losses compared to unmodified graphene and were more stable than the molecular precursors. Finally, a bridging tetrazine derivative was grafted between sheets of graphene oxide to demonstrate that the separation distance between sheets can be maintained while designing new graphene-based materials, including chemically bound, redox structures.
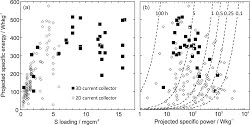
9. M. A. Pope, I. A. Aksay, “Structural Design of Cathodes for Li-S Batteries.” Advanced Energy Materials (2015 - Early View)
Battery technologies involving Li-S chemistries have been touted as one of the most promising next generation systems. The theoretical capacity of sulfur is nearly an order of magnitude higher than current Li-ion battery insertion cathodes and when coupled with a Li metal anode, Li-S batteries promise specific energies nearly five-fold higher. However, this assertion only holds if sulfur cathodes could be designed in the same manner as cathodes for Li-ion batteries. In this critical review, we explore the recent efforts to engineer high capacity, thick, sulfur-based cathodes. We compare various works in terms of capacity, areal mass loading and fraction of conductive additive which are the critical parameters that dictate the potential for a device to achieve a specific energy higher than current Li-ion batteries (i.e., > 200 Wh/kg). While in the majority of cases, we project an inferior specific energy, several promising strategies have the potential to achieve > 500 Wh/kg. We also discuss the challenges associated with the limited cycle-life of these systems due to both the polysulfide shuttle phenomenon and the rapid degradation of the Li metal anode that is experienced at the current densities required to charge high specific energy batteries in a reasonable timeframe.
2014
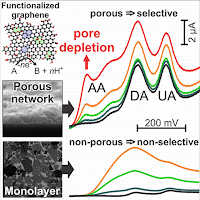
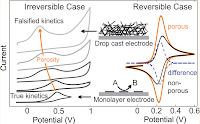
We contrast the performance of monolayer electrodes and thin porous film electrodes of highly reduced functionalized graphene to demonstrate that the introduction of electrode porosity gives rise to strong apparent electrocatalytic effects resulting in vastly improved electrode selectivity. This is despite graphene showing no intrinsic advantage over glassy carbon electrodes when used as a monolayer. The simultaneous electro-oxidation of ascorbic acid, dopamine, and uric acid is used as an experimental model electrolyte system. Our results suggest that a large number of reports claiming the superior surface chemistry of carbon nanomaterials as the reason for outstanding electrochemical characteristics should be revisited considering electrode morphology as a significant contributor to the observed behavior. Our experimental results are supported by numerical simulations explaining the porosity-induced electrode selectivity by the dominance of pore depletion over diffusion-limited currents.
2013
7. M. Liu, C. Punckt, M. A. Pope, A. Gelperin, I. A. Aksay, “Electrochemical Sensing of Nitric Oxide with Functionalized Graphene Electrodes.” ACS Applied Materials & Interfaces 5 (2013) 12624
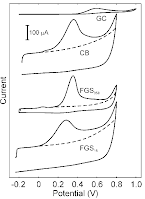
The intrinsic electrocatalytic properties of functionalized graphene sheets (FGSs) in nitric oxide (NO) sensing are determined by cyclic voltammetry with FGS monolayer electrodes. The degrees of reduction and defectiveness of the FGSs are varied by employing different heat treatments during their fabrication. FGSs with intermediate degrees of reduction and high Raman ID to IG peak ratios exhibit an NO oxidation peak potential of 794 mV (vs. 1 M Ag/AgCl), closely matching values obtained with a platinized platinum control (791 mV) as well as recent results from the literature on porous or bio-functionalized electrodes. We show that the peak potential obtained with FGS electrodes can be further reduced to 764 mV by incorporation of electrode porosity using a drop-casting approach, indicating a stronger apparent electrocatalytic effect on porous FGS electrodes as compared to platinized Pt. Taking into consideration effects of electrode morphology, we thereby demonstrate that FGSs are intrinsically as catalytic towards NO oxidation as platinum. The lowered peak potential of porous FGS electrodes is accompanied by a significant increase in peak current which we attribute either to pore depletion effects or an amplification effect due to subsequent electrooxidation reactions. Our results suggest that the development of sensor electrodes with higher sensitivity and lower detection limits should be feasible with FGSs.
Electrodes used in electroanalysis which are based on carbonaceous nanomaterials such as carbon nanotubes or graphene often exhibit large degrees of porosity. By systematically varying the morphology of functionalized graphene electrodes from nearly flat to highly porous, we demonstrate experimentally that minute amounts of electrode porosity have surprisingly significant effects on the apparent reaction kinetics as determined by cyclic voltammetry, both in the reversible and the irreversible regime. We quantify electrode porosity using a coulometric approach and, with the help of numerical simulations, determine the correlation between electrode pore volume and apparent electrode kinetics. We show that in the reversible and quasi-reversible regime, the voltamperometric response constitutes a superposition of thin film diffusion-related effects within the porous electrode and of the standard flat electrode response. For irreversible kinetics, however, we show that diffusive coupling between the electrode and the electrolyte can, under suitably chosen conditions, result in effective electrocatalytic behavior. Confirming past theoretical work by Compton and others, our experiments demonstrate that for a comparison of electroanalytical data obtained with different electrode materials it is not sufficient to only consider differences in the materials’ chemical structure but equally important to take into account differences in electrode morphology.
We use colloidal gels of graphene oxide in a water-ethanol-ionic liquid solution to assemble graphene-ionic liquid laminated structures for use as electrodes in electrochemical double layer capacitors. Our process involves evaporation of water and ethanol yielding a graphene oxide/ionic liquid composite, followed by thermal reduction of the graphene oxide to electrically conducting functionalized graphene. This yields an electrode in which the ionic liquid serves not only as the working electrolyte but also as a spacer to separate the graphene sheets and to increase their electrolyte-accessible surface area. Using this approach, we achieve an outstanding energy density of 17.5 Wh/kg at a gravimetric capacitance of 156 F/g and 3 V operating voltage, due to a high effective density of the active electrode material of 0.46 g/cm2. By increasing the ionic liquid content and the degree of thermal reduction, we obtain electrodes that retain>90% of their capacitance at a scan rate of 500 mV/s, illustrating that we can tailor the electrodes toward higher power density if energy density is not the primary goal. The elimination of the electrolyte infiltration step from manufacturing makes this bottom-up assembly approach scalable and well-suited for combinations of potentially any graphene material with ionic liquid electrolytes.
2011
We present a general method for characterizing the intrinsic electrochemical properties of graphene sheets, such as the specific double-layer capacitance, in the absence of porosity-related artifacts and uncertainties. By assembling densely tiled monolayers of electrically insulating or conductive functionalized graphene sheets onto electrode substrates (gold and highly oriented pyrolytic graphite), we demonstrate our ability to isolate their intrinsic electrochemical response in terms of surface-specific double-layer capacitance and redox behavior. Using this system, the electrochemical properties of various types of graphene can be directly compared without the need to take into account changes in electrode morphology and electrolyte accessibility arising because of varying material properties.
2010
3. C. Punckt, M. A. Pope, J. Liu, Y. Lin, I. A. Aksay “Electrochemical Performance of Graphene as Effected by Electrode Porosity and Graphene Functionalization.” Electroanalysis 22 (2010) 2834
Graphene-based electrodes have recently gained popularity due to their superior electrochemical properties. However, the exact mechanisms of electrochemical activity are not yet understood. Here, we present data from NADH oxidation and ferri/ferrocyanide redox probe experiments to demonstrate that both (i) the porosity of the graphene electrodes, as effected by the packing morphology, and (ii) the functional group and the lattice defect concentration play a significant role on their electrochemical performance.
2. D. H. Wang, R. Kou, D. W. Choi, Z. G. Yang, Z. M. Nie, J. Li, L. V. Saraf, D. H. Hu, J. G. Zhang, G. L. Graff, J. Liu, M. A. Pope, I. A. Aksay, “Ternary Self-Assembly of Ordered Metal Oxide-Graphene Nanocomposites for Electrochemical Energy Storage.” ACS Nano 4 (2010) 1587
Surfactant or polymer directed self-assembly has been widely investigated to prepare nanostructured metal oxides, semiconductors, and polymers, but this approach is mostly limited to two-phase materials, organic/inorganic hybrids, and nanoparticle or polymer-based nanocomposites. Self-assembled nanostructures from more complex, multiscale, and multiphase building blocks have been investigated with limited success. Here, we demonstrate a ternary self-assembly approach using graphene as fundamental building blocks to construct ordered metal oxide−graphene nanocomposites. A new class of layered nanocomposites is formed containing stable, ordered alternating layers of nanocrystalline metal oxides with graphene or graphene stacks. Alternatively, the graphene or graphene stacks can be incorporated into liquid-crystal-templated nanoporous structures to form high surface area, conductive networks. The self-assembly method can also be used to fabricate free-standing, flexible metal oxide−graphene nanocomposite films and electrodes. We have investigated the Li-ion insertion properties of the self-assembled electrodes for energy storage and show that the SnO2−graphene nanocomposite films can achieve near theoretical specific energy density without significant charge/discharge degradation.
2009
1. L.K. Kostanski, M. A. Pope, A. N. Hrymak, M. Gallant, W. L. Whittington, L. Vesselov “Development of Tunable Light Scatting Coating Materials for Fiber Optic Diffusers in Photodynamic Cancer Therapy." Journal of Applied Polymer Science 112 (2009) 1516
To homogenize light emitted from fiber optic diffusers for photodynamic therapy (PDT), novel coating materials with tunable properties were designed. A class VI medical grade UV-curable acrylic resin for biomedical applications was used for the polymer matrix, and titanium dioxide (TiO2) was added to the resin as a scattering agent. UV-curability of coatings containing TiO2 was significantly influenced by the grade and surface treatment of the TiO2 studied. Despite using a free-radical system without inert gas protection, all samples demonstrated considerable thermal postcuring. A novel TiO2 surface treatment methodology was developed based on unsaturated alkoxyorganosilanes combined with unsaturated carboxylic acids for compatibilizing TiO2 for use in the resin material studied. By adjusting the concentration of the surface-modifying agent, the scattering efficiency and UV curability can be controlled by decreasing the effective particle size and enhancing the dispersibility of the powder filler within the resin. This modification will enable fine-tuning of the effective size of TiO2 particles to the wavelength of laser light to be scattered to maximize scattering efficiency in photodynamic cancer therapy and possibly also in other forms of cancer treatment (interstitial laser hyperthermia or interstitial laser photocoagulation).
Patents, patent applications and disclosures
- D. Dabbs, M. A. Pope, I. Aksay, “Electrodes incorporating composites of graphene and selenium-sulfur compounds for improved rechargeable lithium batteries” Princeton University Invention Disclosure 15-3126-1
- M. A. Pope, J. Lettow, I. Aksay, “Embedded monolayers of graphene or graphite platelets as mechanically robust transparent conductors” Vorbeck Materials Corp. Invention Disclosure
- M. A. Pope, V. Alain-Rizzo, J. Lettow, I. Aksay, D. Dabbs, “Batteries incorporating graphene membranes for extending the cycle-life of lithium-ion batteries” U.S. Patent Application #62/012,090
- M. A. Pope, I. A. Aksay, S. Korkut, C. Punckt, “Graphene-ionic liquid composites” U.S. Patent Application #61/600,131
- M. A. Pope, I. A. Aksay, J. D. Roy-Mayhew “Nano-graphene and nano-graphene oxide” U.S. Patent Application #14/017,869
Conference presentations
- Graphene at the Air-Water Interface: Fundamental Studies and Advanced Materials. 3rd Reciprocal WIN-Taiwan Nanotechnology Workshop. August 7th, 2015. (Invited Talk)
- Probing Structure Function Relationships with Graphene Monolayer Electrodes: The Importance of Disorder for Enhanced Electrochemical Performance. M. A. Pope Graphene and Carbon Nanotubes. MRS Spring Meeting. April 10th, 2015. (Talk)
- Development of Graphene-Based Lithium-Sulfur Batteries with High Practical Specific Energy and Power. M. A. Pope. Next-Generation Energy Storage and Conversion Systems. MRS Spring Meeting. April 6th, 2015. (Talk)
- Intrinsic Capacitance of the Functionalized Graphene/Ionic Liquid Interface. D.J. Bozym, M.A. Pope, C. Punckt, I.A. Aksay. Novel Energy Storage Technologies Beyond Li-ion. MRS Spring Meeting. April 24th, 2014. (Talk)
- Intrinsic Capacitance of Functionalized Graphene in Ionic Liquid and Organic electrolytes. D.J. Bozym, M.A.Pope, C. Punckt, I. A. Aksay. Two Dimensional Materials for Energy and Fuel. ACS Spring Meeting, March 19th, 2014. (Talk)
- Beyond the Quantum Capacitance Limit in Graphene-Based Supercapacitors. M.A. Pope, I. A. Aksay. Partner University Workshop, Princeton University. August 27th, 2013. (Invited talk)
- Withdrawal of a Cylinder from an Ellis Fluid. M. Javidi, M.A. Pope, A.N. Hyrmak. Presented at the 16th International Coating Science and Technology Symposium, Midtown Atlanta, GA. September 9-12, 2012. (Talk)
- The Intrinsic Capacitance of Functionalized Graphene Sheets. M.A. Pope, C. Punckt, I. A. Aksay. Carbon Nanotubes, Graphene and Related Nanostructures. MRS Fall Meeting, Boston, MA. November, 29th, 2011. (Poster)
- Electrochemical Double-Layer Capacitors Based on Functionalized Graphene Sheets. M.A. Pope, C. Punckt and I.A. Aksay. Princeton University Chemical and Biological Engineering Graduate Student Symposium. Princeton, NJ. October 28, 2010. (Talk)