We are interested in the solution properties of novel surface active (surfactants) compounds, particularly the so-called gemini or dimeric surfactants. The gemini surfactants (see figure below) have been shown to have a diverse range of solution properties and aggregate morphologies that are dependent upon the structure of the group linking the two surfactant head groups.
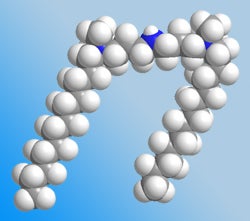
3D Model of the 12-7NH-12 gemini surfactant
Our research has focused on examining what effects occur upon varying the length of a saturated polymethylene spacer group, as well as the effect of substitution of more hydrophilic functional groups within the spacer. It has been shown that substitution of the spacer will, for a variety of reasons, only impact the aggregation properties of the gemini if the spacer group is longer than ~10-12 methylene units in length (Wettig and Verrall, 2001; Wettig, Nowak, and Verrall, 2002; Wettig, Li, and Verrall, 2003).
The cmc values for the gemini surfactants are observed to vary as a function of alkyl tail group length in a manner analogous to that observed for traditional surfactants; namely that the cmc decreases with an increase in alkyl tail length (Kleven's rule). The behaviour of the cmc as a function of the length of the spacer group (assuming a simple polymethylene chain) is more complex and depends upon a combination of steric, electrostatic, and hydrophobic effects that result in specific conformation changes as the surfactant moves from a dispersed to a micellized state.
Recent efforts have focused on the rational design of gemini surfactants for use in non-viral gene therapy vectors as well as for use as bioprobes for the study of the mechanisms of DNA transfection.