Development of new treatments for dermatology, ophthalmology, immunology/vaccines and neurodegenerative diseases using the novel delivery technologies developed in our laboratory:
-
Gene therapy (localized scleroderma, pachyonychia congenita)
Although the genetic bases of most of the common skin diseases and lesions have been elucidated, developing therapeutic strategies based on a knowledge of the underlying genetic mechanisms has been frustratingly slow, largely because of our inability to deliver the therapeutic agents — the genes — into the body safely and reliably.
Skin is the largest and most accessible organ of the body and is an ideal target for gene therapy. Current gene therapy delivery methods include biolistic, microprojectile or direct injection and electroporation. Such methods deliver genes directly into the skin, but they are highly invasive. Alternatively, ex vivo delivery methods could be used. These methods involve removing a skin sample from the patient, culturing skin cells (e.g., epidermal keratinocytes or dermal fibroblasts), genetically engineering such cells in vitro, and then returning the cells as a skin graft to the patient. This procedure, however, can cause scarring and trauma to the patient. Other techniques to delivery genes into the body, such as gene transfer vectors based on viruses, also have many disadvantages.
We are taking a different approach. We are using keratinocytes transfected topically by non-viral delivery systems in the skin as "in situ bioreactors" to produce the desired therapeutic protein for both local treatment and general release of the therapeutic protein into the lymph nodes and the circulatory system. Since delivery is the biggest challenge in gene therapy our main objective is to develop novel non-viral nano-delivery and targeting systems for genes.

Pachyonychia congenita (PC) is an inherited skin disorder. The most common symptoms of PC are focal plantar hyperkeratosis, hypertrophic nail dystrophy, follicular hyperkeratosis, and leukokeratosis of the oral mucosa.
Secondary symptoms may include palmar hyperkeratosis, alopecia or other hair changes, laryngeal involvement, and steatocystoma and pilosebaceous cyst formation. Currently, there is no cure for PC. Patients manage their symptoms in a variety of ways, but no successful drug therapy for this disease has been developed.
The PC Project is a patient support group that helps individuals affected by this disease. The PC Project actively seeks patients with PC, offers help and support, arranges genetic testing to confirm the diagnosis of PC, organizes meetings for patients and researchers, and sponsors research to develop new treatments for the condition.
-
Topical treatments in dermatology and cervical cancer (HPV infections)
Genital warts, one of the most commonly diagnosed sexually transmitted diseases, are caused by the human papillomavirus (HPV). HPV integrates into the epithelial layers of the genital tract, causing proliferated, cauliflower-like lesions. In women, HPV infection can involve vaginal tissues and the cervix, as well as external areas. A major concern about recurrent or resistant HPV infection is the potential of certain strains of the virus (most notably HPV 16 and 18) to cause malignant transformation of untreated but infected areas of the anogenital tract. This can lead to increasing degrees of cervical dysplasia and, ultimately, to cervical cancer.

The National Institute of Allergies and Infectious Diseases (NIAID) estimates that approximately 5.5 million new cases of HPV are diagnosed each year in the U.S., with an estimated 20 million current cases. Worldwide, some 300 million women are carriers of HPV, with a similar number of men also thought to be carriers.
These figures, however, may be just scratching the surface, as the disease is often undiagnosed because patients are too embarrassed to seek medical treatment and because the disease goes untreated as a result of a lack of effective home treatment. Current therapeutic approaches for genital wart removal include conventional and cryosurgery, electrical cautery, laser ablation, or removal with caustic agents such as podophyllin or trichloroacetic acid. These treatments offer temporary relief, but genital warts frequently reoccur because these procedures do not completely eradicate the virus.

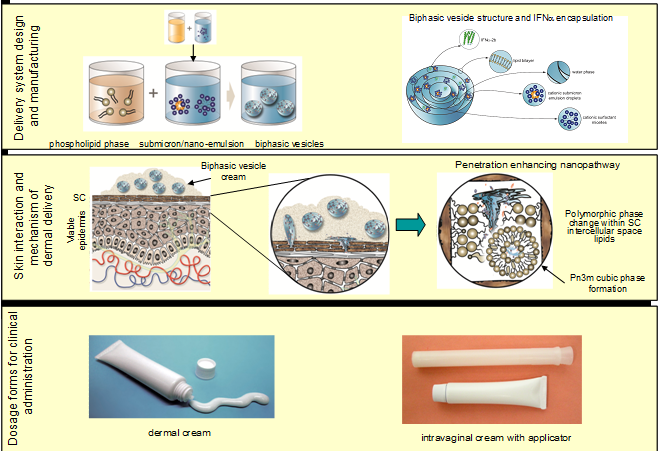
The generally accepted size limit for transdermal delivery of molecules are those that around 500-1000 Da. Above this molecular weight, unassisted permeation of molecules through intact skin is extremely low. Although some early studies demonstrated the feasibility of macromolecule delivery into or through the skin using passive (chemical or delivery system-based) approaches in vitro or in small animals, the first example of successful protein delivery across intact human skin came from my laboratory with the invention of biphasic vesicles. Based on Biphasix™ technology (currently in Phase II clinical trials with IFN-alpha for HPV), this novel delivery system is the first in the world in which a large protein molecule in a pharmaceutical product was administered as a cream applied to the skin, rather than by invasive injections or electrical delivery devices.

-
Targeted drug delivery in neurodegenerative diseases (glaucoma)
Glaucoma is a neurodegenerative disease caused by the slow destruction of retinal ganglion cells and their axons that form the optic nerve. Typically, glaucoma is caused by mechanical injury such as increased intraocular pressure (IOP, normal range 10-21 mmHg). However, local vascular insufficiency at the optic nerve head and increased levels of excitotoxic amino acids such as glutamate may also cause retinal ganglion cell death. Along with the damage to the optic nerve, the entire visual network leading to the visual cortex may be affected. Glaucoma, including primary open-angle glaucoma (POAG) and angle closure glaucoma (ACG), is among the most significant causes of irreversible blindness in the world.
According to recent estimates, the number of people affected worldwide is expected to be over 60 million by the year 2010. We are developing novel delivery systems to stimulate neurite outgrowth and enhance optic nerve regeneration that may one day prevent and treat this devastating disease.

-
Non-invasive vaccine delivery systems
Infectious diseases are still major causes of death and economic loss in both humans and animals. Immunization can prevent many diseases; however, today's vaccines are imperfect, lack optimum efficacy and are painful to administer. Moreover, most vaccines are still being formulated and delivered by injection using delivery devices that haven't changed in decades. There is a great need to develop new delivery methods. Developing non-invasive methods to deliver proteins through impermeable barriers, such as intact skin, will not only greatly facilitate the administration of human and veterinary vaccines, but it will also increase compliance and safety.
The skin contains a network of epidermal Langerhans cells strategically positioned in the epidermis to capture antigens and initiate immune responses. However, the hydrophilic nature and large molecular size of protein antigens, along with the lipophilic character of the skin's stratum corneum, have been regarded as the major barrier in the use of the epidermis as a potential site for vaccine delivery.
We are developing novel delivery vehicles that can penetrate the skin's natural barrier. These non-invasive vaccine delivery systems will have global applicability not just because they would allow needle-free vaccinations but also because the vaccines encapsulated in these systems would have improved stability — e.g., not require cold storage — an obvious advantage in tropical and developing nations.