The Development and Application of 1,2,3-Triiodoarenes in Regioselective Chemical Transformations
Raed
Al-Zoubi
Visiting
Professor
University
of
Waterloo
Wednesday,
January
8,
2019
2:30
p.m.
C2-361
(Reading
Room)
Abstract:
Aryl iodides are versatile and useful compounds in organic chemistry. They are used for the synthesis of remarkable intermediates in agricultural chemicals, pharmaceuticals, and also used as contrast agents in medical applications.[1] Due to the relatively weak nature of the C–I bond, it can be transformed into a plethora of other important organic products, particularly by transition metal catalyzed reactions or by organolithium or organomagnesium intermediates.[2] Although a broad palette of synthetic protocols for iodination of aromatic compounds is available,[3] only a few of them were devoted at the ortho-position.[4]
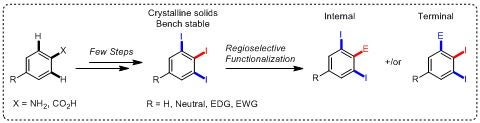
This talk will present three tactics for the synthesis of 1,2,3-triiodoarene derivatives via one-step or two-step protocols from readily available anilines and benzoic acid derivatives.[5] Remarkably, 1,2,3-triiodoarenes are found to be crystalline and bench stable solids. Having only two regiochemically different positions occupied with iodine, the selective functionalization can afford no more than two possible regioisomers, the internal and the terminal products.
A selection of regioselective functionalizations including cascade process will be also presented as well using metal-halogen exchange and transition metal-catalyzed coupling reactions providing the desired iodinated products in good yields, which are valuable building blocks and indeed difficult to make by other means. With other iodo group(s) on the aryl ring, further elaboration can easily be explored. [6]
References:
[1] L. Koehler, K. Gagnon, S. McQuarrie, F. Wuest, Molecules 2010, 15, 2686..
[2] S. J. Blanksby, G. B. Ellison, Acc. Chem. Res. 2003, 36, 255.
[3] E. B. Merkushev, Synthesis 1998, 923; J. R. Hanson, J. Chem. Res. 2006, 277; S. Stavber, M. Jereb, M. Zupan, Synthesis 2008, 1487.
[4] G. C. Clososki, C. J. Rohbogner, P. Knochel, Angew. Chem. Int. Ed. 2007, 46, 7681; S. Usui, Y. Hashimoto, J. V. Morey, A. E. H. Wheatley, M. Uchiyama, J. Am. Chem. Soc. 2007, 129, 151; D. Dolenc, B. Plesnicar, J. Org. Chem. 2006, 71, 8028; E. F. Perozzi, R. S. Michalak, G. D. Figuly, W. H. Stevenson, D. Dess, M. R. Ross, J. C. Martin, J. Org. Chem. 1981, 46, 1049; N. Meyer, D. Seebach, Chem. Ber. 1980, 113, 1304
[5] Al-Zoubi, R. M.; Al-Mughaid, H.; Al-Zoubi, M. A.; Jaradat, K. T.; McDonald, R. Eur. J. Org. Chem. 2015, 5501; R. M. Al-Zoubi, H. A. Futouh, R. McDonald, Aust. J. Chem. 2013, 66, 1570-1575; Al-Zoubi, R. M.; Al-Mughaid, H.; McDonald, R. Aust. J. Chem. 2015, 68, 912-918.
[6] Al-Zoubi, R. M.; Al-Zoubi, M. S.; Abazid, A. H.; McDonald, R. Asian J. Org. Chem. 2015, 4, 359; Al-Zoubi, R. M.; Al-Jammal, W. K.; El-Khateeb, M. Y.; McDonald, R. Eur. J. Org. Chem. 2015, 3374; R. M. Al-Zoubi, A. Ibdah, W. K. Al-Jammal, M. S. Al-Zoubi, A. A. Almasalma, R. McDonald, Synthesis 2018, 50, 384-390; R. M. Al-Zoubi, M. S. Al-Zoubi, K. T. Jaradat, R. McDonald, Eur. J. Org. Chem. 2017, 5800-5808;