If you would like to see more information on this case study, click here!
You can request this case study and a WCDE staff member will get back to you.
The Chemical Engineering Reaction Engineering Lab, at the University of Waterloo, studies viable energy sources to replace the dependency on petroleum. The main focus is to develop alternative sources of hydrogen fuel using various methods. Hydrogen can be produced from natural gas by dry reforming and partial oxidation of methane. In some cases it is attractive and viable to convert hydrogen to liquid fuels such as methanol. Methanol is in demand as feedstock for chemical production, biodiesel production, and to generate electricity. The Reaction Engineering Lab studied through simulation a methanol production plant from landfill gas at a company’s request using the model for a methanol production plant as illustrated in Figure 1.
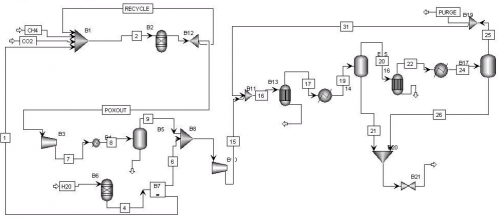
Muhammad Abduhu, a co-op student from the University of Waterloo, was asked to investigate ways to optimize and improve this simulation for the production of methanol by analyzing the energy requirements of each component of the system.
The teaching objective is to facilitate decision making skills for process flowsheet design and optimization through simulation. The case study is intended to introduce students to AspenPlus software. The case can potentially be used in CHE 290/291 (Chemical Engineering computer laboratory), or CHE 480 (Process Analysis and Design) courses.
If you would like to see more information on this case study, click here!
You can request this case study and a WCDE staff member will get back to you.
Contact Waterloo Cases in Design Engineering
Steve Lambert
Tel: (519) 888-4728
Email: steve@uwaterloo.ca
The University of Waterloo acknowledges that much of our work takes place on the traditional territory of the Neutral, Anishinaabeg, and Haudenosaunee peoples. Our main campus is situated on the Haldimand Tract, the land granted to the Six Nations that includes six miles on each side of the Grand River. Our active work toward reconciliation takes place across our campuses through research, learning, teaching, and community building, and is co-ordinated within the Office of Indigenous Relations.