Waterloo Pharmacy student research lands cover spot in Medicinal Chemistry Research
Volume 35 of the Medicinal Chemistry Research journal cover features art compiled by University of Waterloo School of Pharmacy student Rahul Karuturi (PhD’25) for their research, Synthesis and evaluation of symmetric (1,4-diazepane-1,4-diyl)bis(phenylmethanone) derivatives as amyloid-beta aggregation inhibitors.
Alzheimer's Disease (AD) is a complex and devastating neurodegenerative disorder marked by the abnormal folding and accumulation of amyloid proteins in the brain. In particular, amyloid-beta peptides (Aβ42 and Aβ40) misfold, aggregate, and form toxic plaques that disrupt communication between neurons and ultimately lead to brain cell death. These pathological changes progressively impair memory, cognition and daily functioning. AD has no cure and current medications only relieve a patient’s symptoms and do not target the underlying cause. AD is a growing global health challenge, affecting millions worldwide, with numbers expected to rise sharply in the coming decades. In Canada alone, around 733,000 people are currently living with dementia and projected to reach 1 million by 2030.
The recently approved monoclonal antibody based medications are promising but are very expensive and not suitable for all patients; they may result in serious side effects and are administered through an injection. As part of his PhD research Karuturi investigated a novel class of 1,4-diazepane derivatives as amyloid aggregate inhibitors, which prevent the formation of these protein clumps and are cost-effective with lower risk of serious side effects. These novel molecules functions as a dual inhibitor, targeting both Aβ42 and Aβ40, exhibits antioxidant activity, and can cross the blood-brain barrier to provide neuroprotection to brain cells.
“It is anticipated that these findings will provide novel drug design concepts to discover anti-AD agents, leading to therapies that slow or prevent disease progression, potentially improving patients’ quality of life, reducing caregiver stress, lowering healthcare costs and lessening the societal and economic impact of the disease,” says Karuturi.

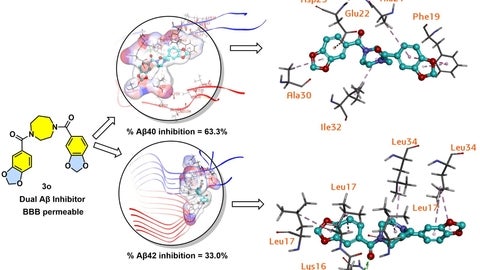
To illustrate this research Karuturi designed the cover image using Discovery Studio Structure-Based Design and ChemDraw to highlight the key molecular interaction of the novel 1,4-diazepane derivative and created a detailed representation and its interactions with Aβ42/40 peptides.
The ball and stick cartoon represent 1,4-diazepane derivative designed to block two different biological targets at the same time, in this case the Aβ42/40 peptides. The ribbon diagram represents the amyloid peptides that 1,4-diazepane derivative targets to prevent aggregation in a patient’s brain with AD. The cover was designed to visually depict the mechanism, showing how the novel molecule interacts with amyloid aggregates. Next step in this research includes optimizing the structural parameters to obtain molecules with superior activity and evaluate them in different models of Alzheimer’s disease.
Karuturi worked in the laboratory of Dr. Nekkar at the School of Pharmacy and the research received financial support from NSERC-Discovery, the Canada Foundation for Innovation (CFI-JELF), the Ontario Research Fund (ORF), the Pooler Charitable Foundation, and the University of Waterloo, School of Pharmacy.